New research collaboration on liver health with the University of Vienna
2026-03-13
We are excited to announce the launch of a strategic research collaboration with the University of Vienna and three of their leading university research groups in the Department of Pharmaceutical Sciences to accelerate the development of innovative therapies for Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)—one of the world’s fastest growing liver health challenges.
MASLD has emerged as a major global health burden, affecting millions of people and driving significant increases in morbidity, mortality, and healthcare costs. With prevalence closely linked to obesity, diabetes, and metabolic dysfunction, MASLD represents a critical unmet medical need requiring new therapeutic strategies.
Our joint research initiative as part of our global Schwabe Phyto Innovation Challenge initiative will focus on the identification of natural products targeting thyroid and bile acid receptors, two highly promising pathways at the forefront of next generation MASLD treatment.
As a pharmaceutical company specializing in evidence based phytomedicine, we are proud to bring our deep methodological expertise in plant based drug development into this collaboration. By combining our capabilities with the scientific excellence of our academic partners, we aim to unlock new therapeutic potential in an area where innovation is urgently needed.
This partnership underscores our commitment to driving translational research, advancing novel mechanisms of action, and strengthening our role as a pioneer in modern phytopharmaceutical science.
Picture (left to right): Prof Johannes Kirchmair, Uday Abu-Shehab, Eva Waltenberger, Dr. Martin Lehner (Schwabe Group), Prof. Judith Rollinger, Katharina Riffelsberger, Patrik Schwarz., Prof. Verena M. Dirsch, Dr. Dirk Bredenbröker (Schwabe Group)
Treatment effects of Ginkgo biloba extract EGb 761® in patients with chronic tinnitus
2026-02-17
A study recently published by Schwabe investigated the effects of the Ginkgo biloba extract EGb 761® in chronic tinnitus. A total of 170 patients took 120 mg of the extract twice daily for 24 weeks.
Results:
- After 24 weeks, patients had significantly fewer tinnitus symptoms.
- People who had high levels of anxiety or stress at the start of the study improved particularly strongly.
- Patients with normal hearing also benefited more.
The study demonstrated that EGb 761® can help reduce symptoms of chronic tinnitus. It appears to be especially beneficial for people with normal hearing as well as those experiencing anxiety or stress.
The publication
Lisowska, G.; Urban, I.; Skarżyński, P.H.; Schlaefke, S.; Brueggemann, P.; Mazurek, B. Ginkgo biloba Extract EGb 761® in Patients with Chronic Tinnitus: Treatment Effects and Effect Modifiers. J. Clin. Med. 2026, 15, 87. Doi 10.3390/jcm15010087.
can be freely accessed here: Ginkgo biloba Extract EGb 761® in Patients with Chronic Tinnitus: Treatment Effects and Effect Modifiers
Walmer Primary School is our 2026 "School in Africa"
2026-02-13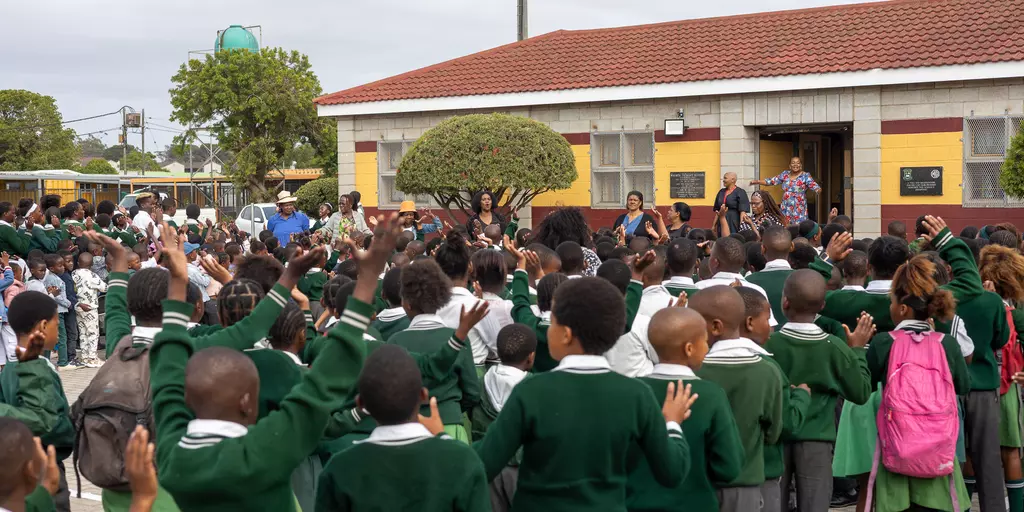
Education creates opportunities – starting from the earliest years
In 2026, the Umckaloabo Foundation will support the Walmer Primary School in the Walmer Township (near Port Elizabeth) in South Africa as part of the
The

A Community Hall that Transforms School and Township Life
Together with our project partner Masifunde, we aim to enable the construction of a much‑needed community hall on the school grounds. Until now, school assemblies have taken place outdoors – under the burning sun or in pouring rain. The new hall will not only improve daily life for the roughly 1,800 students but also strengthen the sense of community: outside school hours, the hall will be open to residents of the township.
Other project partners are also on board, funding the creation of a traffic education area as well as the construction of new classrooms. This is how a greater whole comes together!

Running for a Better Future
The
This project is about far more than new buildings: it strengthens the entire township, fosters community and creates real opportunities for future generations. It also deepens the connection between the school and the Masifunde Education Centre.
This is how positive change becomes possible. Because education changes lives.
Schwabe Group to Lead Innovation at the European Herbal Health Products Summit
2026-02-10
On 25 February 2026, key stakeholders from across Europe will come together in Brussels for the European Herbal Health Products Summit.
The summit will provide a high‑level platform to discuss the role of herbal medicinal products in the EU, their economic and societal relevance, and the regulatory challenges and opportunities shaping the future of the sector.
Organised by Pharma Deutschland in cooperation with the International Society for Medicinal Plant and Natural Product Research (GA), the event brings together policymakers, regulators, industry representatives and scientific experts for an open and forward‑looking exchange.
We at the Schwabe Group are extremely proud to contribute to the event with our own input on innovation, sharing perspectives on how scientific excellence and innovative approaches can help further strengthen herbal medicinal products in Europe.
Together for biodiversity at Schwabe
2026-02-03
Hands-on commitment at Terra Medica
At the end of January, a dedicated group of employees gathered at the Schwabe medicinal plant cultivation site,
Learning and doing—side by side
Together, the Schwabe colleagues filled the large wooden frame with bamboo and reed tubes of various sizes. It was meticulous work, but also an opportunity to learn. Insect expert Hans-Joachim Greiler joined the activity, guiding the construction and sharing insights and surprising facts about wild bees and their vital role in our ecosystem. A great thank you for that!


For birds, too
While one team focused on the bee nesting aid, another group cleaned and prepared the many bird nesting boxes for the new season and recorded, which bird species call Terra Medica their home.


Eighteen of the 35 nesting boxes were occupied, mainly by titmice. The volunteer team was in for a surprise when they opened one of the large bat boxes. Contrary to expectations, it did not contain any traces of bats, but instead nests built by various other animals — ranging from several bird nests to a hornet’s nest and even a bumblebee nest. And last but not least, the team discovered a dozen field wasps that were using the old hornet cells as winter quarters.


Ready for the “nest generation”
The finished nesting aid is now in place at

Dr. Traugott Ullrich to Lead New Business Unit: Innovation & Market Access at Schwabe Group
2026-02-02
As part of our ongoing organizational development, the Schwabe Group is combining Research & Development and Global Regulatory Affairs into a new business unit: Innovation and Market Access.
This strategic move aims to:
✅ Secure and expand the market presence of our products
✅ Drive customer- and market-focused development of new health solutions in close alignment with Marketing and Sales
✅ Ensure rapid market access for innovative offerings
Additionally, the new unit will coordinate the Schwabe Group’s national and European pharma policy activities to maintain and strengthen optimal regulatory and political conditions for our business model.
We are pleased to announce that Dr. Traugott Ullrich will lead this new division as EVP Innovation & Market Access, effective February 1, 2026, reporting directly to Olaf Schwabe, CEO of the Schwabe Group.
Dr. Ullrich combines profound medical knowledge with extensive commercial expertise. Working for many years successfully as a Board Member in national and European Pharmaceutical Associations (AESGP, Pharma Deutschland) he can complement this with a sound understanding of the political and regulatory framework thus shaping the future of our health solutions.
New Phase IV evidence for Menthacarin® published
2026-01-19
We are thrilled to announce that our latest study results on Menthacarin® have now been published in the scientific journal Wiener Medizinische Wochenschrift.
The study followed 126 patients with functional dyspepsia or irritable bowel syndrome–like symptoms over the course of an eight-week therapy with our peppermint oil–caraway oil combination Menthacarin® (2 × 1 capsule per day).
Key outcomes
• Abdominal symptoms like pain, cramps or bloating showed significant improvement
• Symptom days per week almost halved
• More days with normal stool consistency and fewer episodes of incomplete bowel movements
• Marked improvement in health‑related quality of life
• Excellent tolerability profile confirmed
These trial data underline the patient-relevant benefits of our evidence-based phytotherapeutic for disorders of gut–brain interaction.
🔗 Read the full paper: Treatment of disorders of gut–brain interaction with peppermint oil and caraway oil combination Menthacarin | Wiener Medizinische Wochenschrift
Schwabe Group’s CO2 emissions – 24% lower than baseline year
2025-12-15
The Schwabe Group’s total emissions in Scope 1 and 2 in 2024 were 37,266 t of CO₂ (market-based approach). In 2021, our baseline year, our global carbon emissions in Scope 1 and 2 were still 48,871 to. So the development is extremely positive: in comparison 2021, the Schwabe Group reduced the Scope 1 and 2 numbers in 2024 by approximately 24% and eliminated 11,605 tons of CO₂e!
By doing so, we have taken a large step closer to our mid-term goal of cutting our CO₂ emissions in half by 2030. To continue decreasing our carbon footprint, we have started various projects, among them: a geothermal project in the US and a large solar-panel system in India.
Top 5 stayed the same
The largest sources of CO₂ emissions in the Schwabe Group are still the sites where production and/or extraction takes place: the US, Germany, India, Ireland and Mexico, which together are responsible for 92% of carbon emissions in the Group in 2024.
Society for Phytotherapy: Schwabe's Stephan Köhler elected as the new Chair
2025-11-20
At yesterday’s meeting of the Advisory Board of Corporate Members of the Society for Phytotherapy at Pharma Deutschland in Bonn, the board held its scheduled session and addressed key topics for the future of phytotherapy.
A central point of the meeting was the election of the entire Advisory Board, with our colleague Stephan Köhler, Director Global Pharmacovigilance at the Schwabe Group, chosen as the new Chair. Congratulations, Stephan, on taking on this important responsibility!
We, as the Schwabe Group, are truly delighted that you have taken on this role to further drive innovation and uphold the highest quality standards in phytopharmaceuticals worldwide.
In addition to the inaugural session, the board focused on comments on European guidelines, including the Reflection Paper on Patient Experience Data—a document that will shape future regulatory expectations and strengthen patient-centric approaches across the healthcare landscape.
Caption: The Advisory Board, from left to right: Dr. Inga Trompetter, Prof. Dr. Freimut Schliess, Dr. Barbara Steinhoff, Dr. Stephan Köhler, Dr. Christoph Theurer, Prof. Dietmar Kammerer, Dr. Nico Symma.
When volunteering pays off ...
2025-11-03
Schwabe Group awards €1,000 to six employees engaged in volunteer work
The Schwabe Group supports the voluntary and charitable commitment of its employees: In autumn 2025, six Schwabe employees each received a Schwabe Community Grant of €1,000 for the projects or initiatives in which they are personally involved.
This time, the awardees are Schwabe employees who are active in areas such as child and youth services, hospice care, and cultural promotion. The Schwabe Group’s Community Grant serves both as recognition and encouragement for these dedicated individuals.
In awarding these Grants, the Schwabe Group extends its social commitment beyond the scope of the
Four examples of voluntary and community engagement by Schwabe employees who received a Community Grant:



